The Scribble family in cancer: twentieth anniversary.
Santoni MJ, Kashyap R, Camoin L, Borg JP


The Marseille Cancer Research Center celebrates its 50th anniversary ! -
Our team explores molecular circuits operating in solid tumors, focusing particularly on components of signaling networks that represent novel vulnerabilities still poorly exploited, notwithstanding their key role in cancer. We investigate their mode of action in cancer cells and the mechanisms associated, as well as their implication in crosstalks operating in the tumor microenvironment between distinct cell types. Studies are performed through complementary – single to complex – strategies. At molecular levels, we combine biochemical with -omics approaches. At cellular levels, we employ in vivo and ex vivo approaches with sophisticated cancer mouse models. We widen our studies through interdisciplinary strategies in collaborative settings, including computational biology, mathematical modeling, and physics. We explore these circuits on poor prognostic cancers, at the root of tissue homeostasis destabilization, tumor initiation, progression, and how these circuits correspond to putative vulnerabilities to exploit for targeted therapies.
The team topics are centered on two main axes:
We expand our understanding at functional, mechanistic, and molecular levels. Importantly, these signals represent new putative targets for anticancer treatments, for which we are developing targeting agents.
Among aspects we explore:
This axis aims at exploring the implication of new candidate genes/mechanisms in the crosstalk between cancer cells and their microenvironment, particularly with immune cells. It is based on the combination of datasets and samples from patients with ex vivo (organoids/tumoroids), single cell and spatial-omics, and in vivo cancer models.
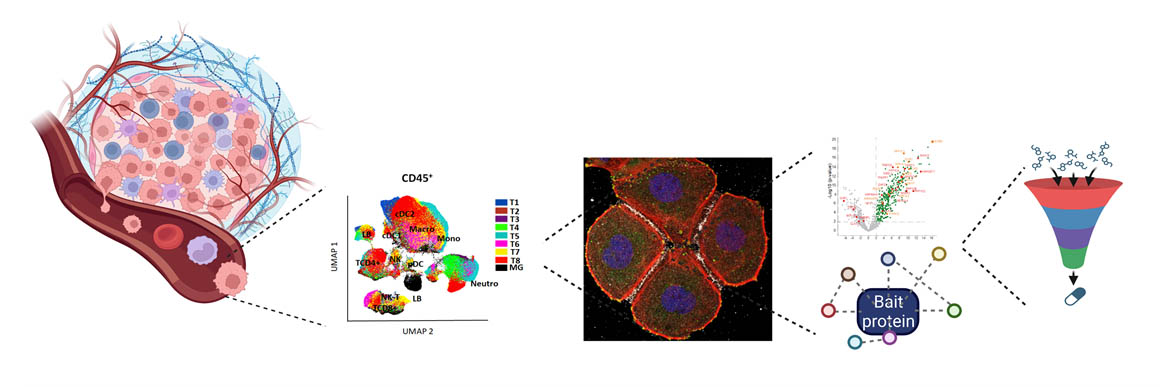 From left to right: Schematic representation of a solid tumor, Cellular clustering by UMAP, Cell immunofluorescence, Protein interaction (Volcano plot and network), Drug identification.
From left to right: Schematic representation of a solid tumor, Cellular clustering by UMAP, Cell immunofluorescence, Protein interaction (Volcano plot and network), Drug identification.
This axis focuses on emerging signaling pathways in cancer, whose functions and mechanisms of action remain largely unknown. These pathways may represent potential new targets for anticancer treatments, for which we are developing targeting agents. Among the aspects we are exploring:
a) Mechanistic relationships between multiple signaling networks;
b) Regulation of biological processes in cancer cells (tumor initiation, cell dissemination, collective cell migration, treatment resistance, etc.) and their microenvironment;
c) Design of optimized strategies for the generation of blocking agents.
The WNT/Planar Cell Polarity (PCP) pathway is the most recently described branch of WNT signaling and is strongly implicated in both early- and late-stage cancer development. Over the years, we have carried out fundamental work on the mode of action and role of WNT/PCP signaling in aggressive cancers, and recently reviewed the most promising strategies currently designed to target its membrane or sub[1]membrane components. Furthermore, we have uncovered that ADAMTSL5, a secreted protein which interacts with poorly understood molecular partners, is overexpressed in various types of cancer with poor prognosis, representing a new therapeutic target. Our studies are predominantly focused on the following signaling molecules.
1.1. VANGL2 is a receptor of the WNT/PCP pathway with four transmembrane regions, two extracellular loops and two intracellular regions. We found that overexpression of VANGL2 in triple-negative breast cancer (TNBC) correlates with poor prognosis. The mode of action of VANGL2 still remains enigmatic despite the data accumulated by various teams including us. We have recently copurified VANGL2 with a G protein-coupled adhesion receptor (GPCR). A transcriptomic analysis of a large cohort of TNBC patients from the Institut Paoli-Calmettes (IPC) and public data showed an association between this GPCR and decreased metastasis-free survival, with a synergistic effect of VANGL2.
We are currently:
1) studying the molecular organization of the VANGL2/GPCR pathway complex and its contribution to the aggressiveness of TNBC;
2) developing antibodies capable of detecting the active form of the GPCR in tissue sections from TNBC (Tissu MicroArray, > 250 biopsies, IPC) for clinical correlation studies;
3) dissecting the GPCR downstream signaling and the impact of VANGL2 on the latter in normal and breast cancer cells using different energy transfer methods (BRET/FRET);
4) generating pharmacological modulators blocking the pro-tumorigenic functions of the GPCR.
1.2. MINK1, a serine-threonine kinase of the WNT/PCP pathway, promotes tumor growth and dissemination of breast cancer cells to distant organs (lung, liver). Members of the MINK1-related protein kinase family also regulate WNT, HIPPO, and AKT signaling. We are:
1) developing MINK1 inhibitors for therapeutic use;
2) characterizing biomarkers capable of predicting the efficacy of MINK1 inhibitors;
3) dissecting MINK1 signaling using mass spectrometry approaches;
4) evaluating MINK1 inhibitors in a unique TNBC genetic mouse model, in Patient-Derived Xenografts (PDX), and in TNBC tumoroids.
1.3. PTK7 is a pseudo-tyrosine kinase receptor overexpressed in many types of cancer, associated with poor prognosis and metastatic spread. PTK7 plays a dual role in the WNT/PCP and WNT/β-catenin pathways by binding directly to β-catenin. This receptor is currently targeted by an antibody-drug Project(s) - ENGLISH A more detailed text where you can provide information about your project(s). We have recently developed small molecules capable of inhibiting the interaction between PTK7 and β-catenin. We are currently:
1) deploying a holistic approach to the search for inhibitor molecules by studying structure-activity relationships;
2) assessing the efficacy of selected compounds in vitro and in vivo in colorectal cancer.
1.4. ADAMTSL5 is a gene we have identified through -omics approaches, which has never been linked to cancer prior to our studies. ADAMTSL5 is a secreted glycoprotein, lacking a catalytic activity, and characterized by a Netrin domain located in the C-terminal region that is not present in other family members. We have documented its involvement in hepatocellular carcinoma (HCC) and shown that its inhibition leads to a loss of a hybrid epithelial-mesenchymal state, characterizing aggressiveness, accompanied by the acquisition of a fibroblastic-like state. Targeting ADAMTSL5 expression leads to a depletion of several oncogenes, including receptors with tyrosine kinase (RTKs), interferes with the tumoral properties of cancer cells, and confers sensitivity to drugs used in the clinic. We are:
1) validating ADAMTSL5 as a biomarker;
2) deciphering the protein complex associated with ADAMTSL5 and its mechanism of action;
3) studying how ADAMTSL5 impacts the tumor microenvironment, in particular the immune system;
4) generating agents for diagnosing and targeting ADAMTSL5 (e.g., antibodies, aptamers,…).
This axis aims to explore the involvement of new candidate genes/mechanisms in the crosstalk between cancer cells and their microenvironment, in particular with immune cells. It is based on the combination of patient datasets and samples with ex vivo (organoid/tumoroid), single-cell and spatial, and in vivo cancer models.
2.1. Focusing on TNBC, we use unique in vivo cancer models we have generated to explore the composition of different immune cell types present in the tumor microenvironment and their interaction with cancer cells. We reconstructed and targeted these interactions in vivo by developing orthotopic models in syngeneic mice with an intact immune system. Results are systematically compared with those found in patients. We are exploring longitudinally how immune cell remodeling occurs in relation to the type of treatment. Studies are performed through dynamic rather than static analyses of the immune cell composition, to uncover how immune remodeling in the tumor microenvironment may influence the selection of relevant immunotherapies for combinatorial treatments. We anticipate that the acquisition of this knowledge could help to design new combinations of anticancer agents and immunotherapies.
2.2. We have preliminary data indicating that early preneoplastic lesions are characterized by cellular and molecular features that diversify their behavior as quiescent versus evolving lesions. Focusing on liver cancer, we extended these studies using unique mouse models of spontaneous tumorigenesis, recapitulating the molecular and the tumor onset heterogeneity. By applying a powerful imaging system based on photon-counting computed tomography (PC-CT; established by our collaborators at the CPPM), we are able to longitudinally track endogenous tumors over time. Quiescent and evolving lesions are processed for spectral cytometry and single cell RNAseq, to uncover the molecular and cellular features that diversify evolving versus quiescent behavior. We assess the robustness of the results functionally using ex vivo and in vivo models, and clinically by analyzing patient data and samples.
2.3. Role of PTK7 in tumor-host interactions. We recently discovered that PTK7 is expressed in dendritic cells (DCs), particularly Langerhans cells (skin, cutaneous lymph nodes) in mice. In addition, PTK7 expression was also observed in tumor-infiltrating monocytes, pDCs, cDC2 and cDC1 in murine models of melanoma and breast cancer, suggesting that PTK7 may be modulated by the tissue microenvironment. Combining a genetic mouse model with multi-parametric -omics data, we are:
1) investigating the physiological function of PTK7 in dendritic cells (DCs);
2) assessing the role of PTK7-expressing DCs in breast cancer.
We have identified PTK7 expression in colonic fibroblasts in murine and human colorectal cancer (CRC). We have generated PTK7-deficient mice in a subset of colonic fibroblasts and revealed susceptibility to acute colitis and probable tumorigenesis. These results indicate an essential role for PTK7 in the crosstalk between colonic epithelium and fibroblasts. We are:
1) documenting the PTK7-dependent landscape and the crosstalk between colonic epithelium and fibroblasts during tissue homeostasis;
2) molecularly and functionally characterizing the different cellular compartments regulated by PTK7 using relevant mouse models and subtype-annotated human organoids;
3) evaluating PTK7 binding partners in epithelial and fibroblastic compartments using a PTK7-BirA* "knock-in" mouse model of colon carcinogenesis;
4) deciphering the contribution of PTK7-expressing fibroblasts to colon cancer tumorigenesis;
5) validating PTK7 expression and alterations identified in mice as prognostic markers in colon cancer patients.
Santoni MJ, Kashyap R, Camoin L, Borg JP
Daulat AM, Finetti P, Revinski D, Silveira Wagner M, Camoin L, Audebert S, Birnbaum D, Kodjabachian L, Borg JP, Bertucci F
Rochigneux P, Lisberg A, Garcia A, Granjeaud S, Madroszyk A, Fattori S, Gonçalves A, Devillier R, Maby P, Salem N, Gorvel L, Chanez B, Gukasyan J, Carroll J, Goldman J, Chretien AS, Olive D, Garon EB
Montserrat-Gomez M, Gogl G, Carrasco K, Betzi S, Durbesson F, Cousido-Siah A, Kostmann C, Essig DJ, Strømgaard K, Østergaard S, Morelli X, Trave G, Vincentelli R, Bailly E, Borg JP
Walton A, Thomé V, Revinski D, Marchetto S, Puvirajesinghe TM, Audebert S, Camoin L, Bailly E, Kodjabachian L, Borg JP